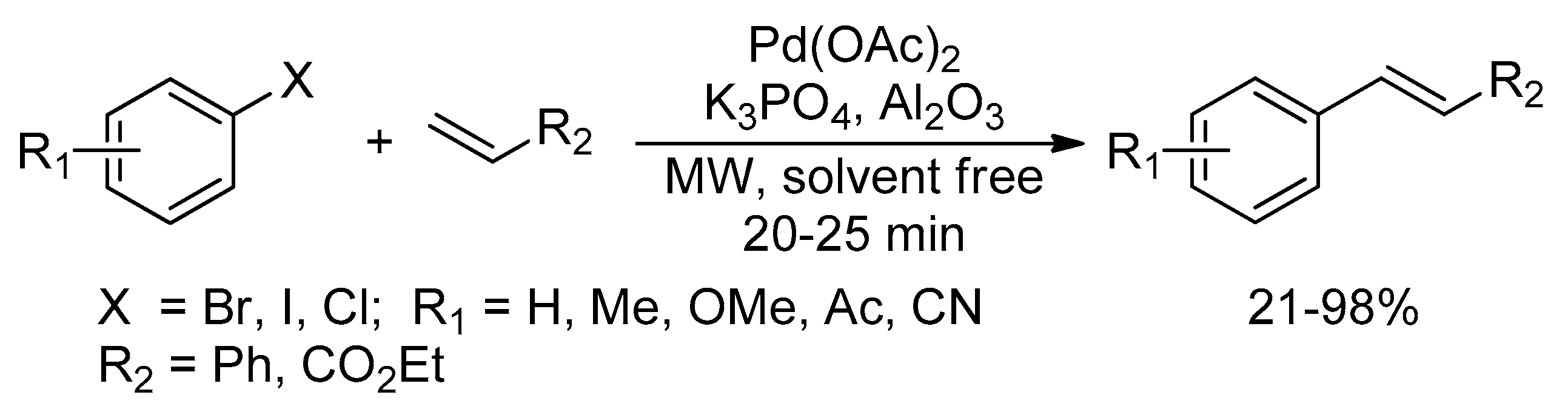New trends and applications in cyanation isotope chemistry - Derdau - 2018 - Journal of Labelled Compounds and Radiopharmaceuticals - Wiley Online Library
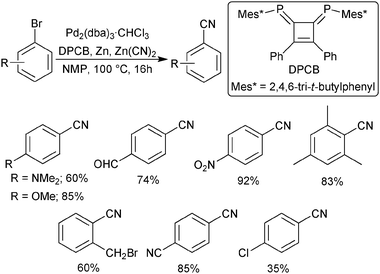
Recent advances and prospects in the palladium-catalyzed cyanation of aryl halides - RSC Advances (RSC Publishing) DOI:10.1039/D0RA05960A

An overview of microwave assisted cyanation reactions - Fairoosa - 2021 - Applied Organometallic Chemistry - Wiley Online Library

Controllable cyanation of carbon-hydrogen bonds by zeolite crystals over manganese oxide catalyst | Nature Communications

New trends and applications in cyanation isotope chemistry - Derdau - 2018 - Journal of Labelled Compounds and Radiopharmaceuticals - Wiley Online Library
![A mild and efficient palladium-catalyzed cyanation of aryl chlorides with K4[Fe(CN)6]. | Semantic Scholar A mild and efficient palladium-catalyzed cyanation of aryl chlorides with K4[Fe(CN)6]. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/861f9c3deb3dc91f7afcf7091bed27b2ed59c45c/2-Figure1-1.png)
A mild and efficient palladium-catalyzed cyanation of aryl chlorides with K4[Fe(CN)6]. | Semantic Scholar

PDF) ChemInform Abstract: Synthesis of Phthalonitriles Using a Palladium Catalyst | zafar Iqbal - Academia.edu
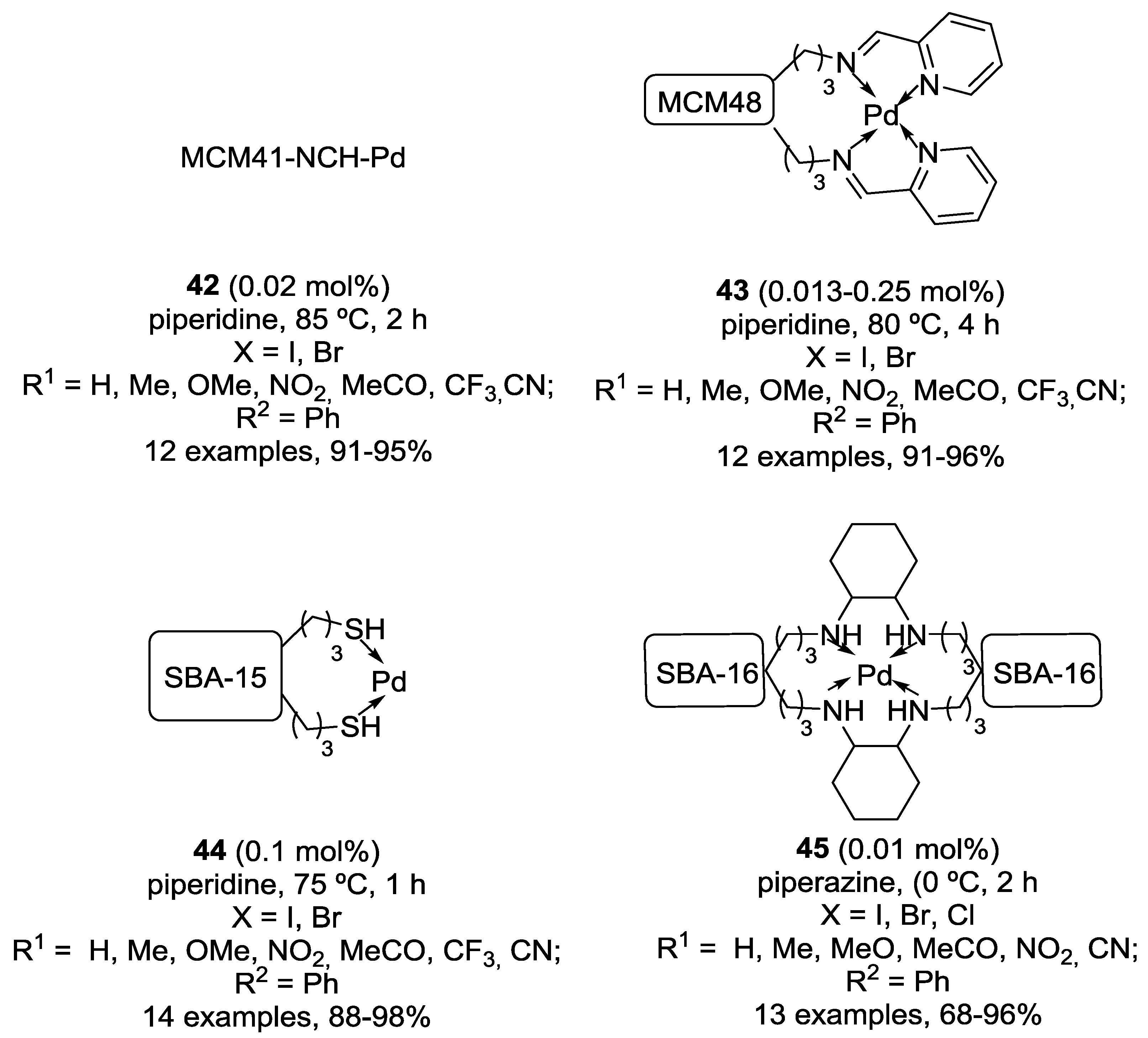
Catalysts | Free Full-Text | Solid-Supported Palladium Catalysts in Sonogashira Reactions: Recent Developments | HTML

A General, Practical Palladium‐Catalyzed Cyanation of (Hetero)Aryl Chlorides and Bromides - Senecal - 2013 - Angewandte Chemie International Edition - Wiley Online Library

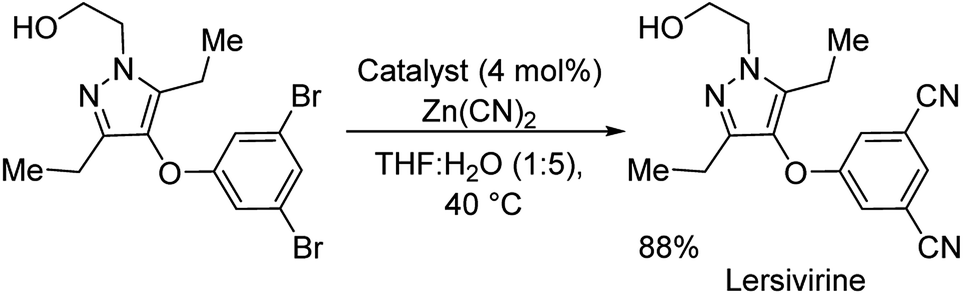
General and Mild Nickel-Catalyzed Cyanation of Aryl/Heteroaryl Chlorides with Zn(CN)2: Key Roles of DMAP. | Semantic Scholar

Microwave-enhanced cyanation of aryl halides with a dimeric ortho-palladated complex catalyst | SpringerLink
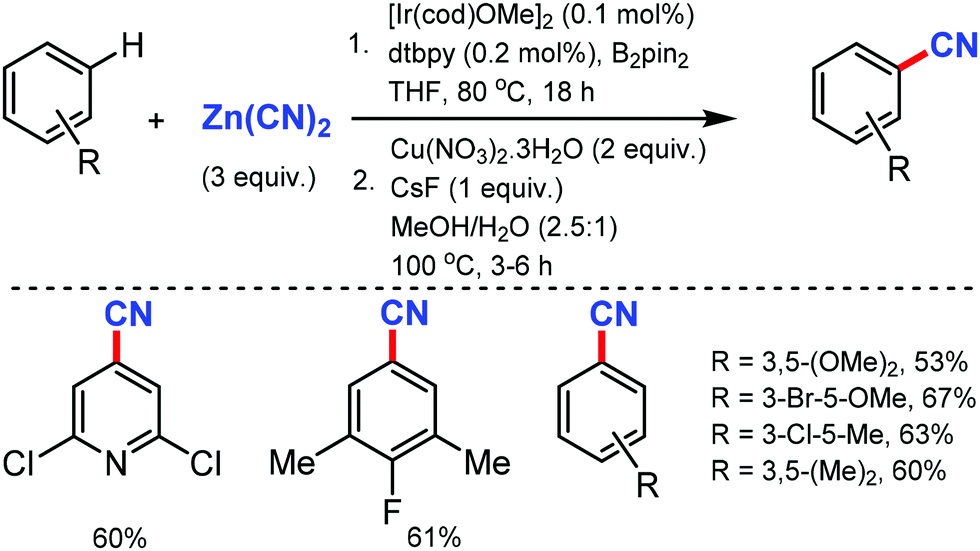
C–CN bond formation: an overview of diverse strategies - Chemical Communications (RSC Publishing) DOI:10.1039/D0CC07783F

A general, practical palladium-catalyzed cyanation of (hetero)aryl chlorides and bromides. | Semantic Scholar