Pharmaceutics | Free Full-Text | Combining Therapeutic Drug Monitoring and Pharmacokinetic Modelling Deconvolutes Physiological and Environmental Sources of Variability in Clozapine Exposure | HTML

Development of a population pharmacokinetic model of olanzapine for Chinese health volunteers and patients with schizophrenia | BMJ Open

Establishing Best Practices and Guidance in Population Modeling: An Experience With an Internal Population Pharmacokinetic Analysis Guidance - Byon - 2013 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library

CHARACTERIZATION OF THE TIME-VARYING CLEARANCE OF RITUXIMAB IN NON-HODGKIN'S LYMPHOMA PATIENTS USING A POPULATION PHARMACOKINETIC ANALYSIS METHODS INTRODUCTION. - ppt download
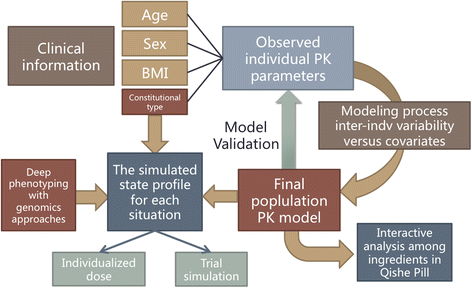
Population pharmacokinetic modeling of the Qishe pill in three major traditional Chinese medicine-defined constitutional types of healthy Chinese subjects: study protocol for a randomized controlled trial | Trials | Full Text
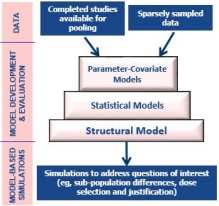
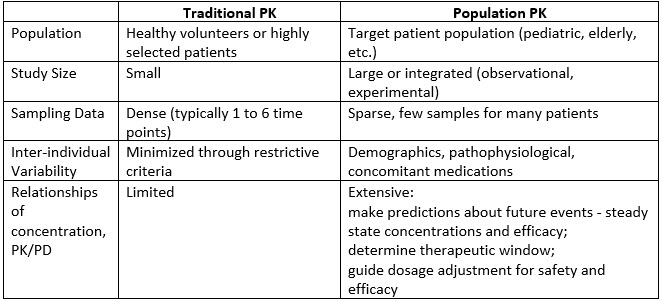
Session 1: An Introductory Workshop in Population PK/PD Data Analysis with NONMEM® (In Person) - School of Pharmacy and Pharmaceutical Sciences - University at Buffalo

Model-Informed Drug Development, Pharmacokinetic/Pharmacodynamic Cutoff Value Determination, and Antibacterial Efficacy of Benapenem against Enterobacteriaceae | Antimicrobial Agents and Chemotherapy

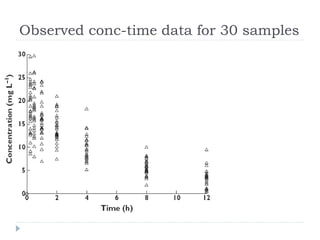
PopPK analysis: prediction-corrected visual predictive check for (a)... | Download Scientific Diagram

Population Pharmacokinetics Analysis of Quetiapine Extended-release Formulation in Japanese Patients with Bipolar Depression - Clinical Therapeutics

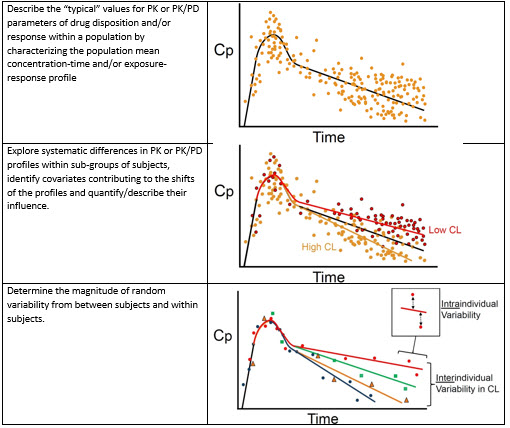
Overview of PopPK analysis. IIV, inter-individual variability; PopPK,... | Download Scientific Diagram
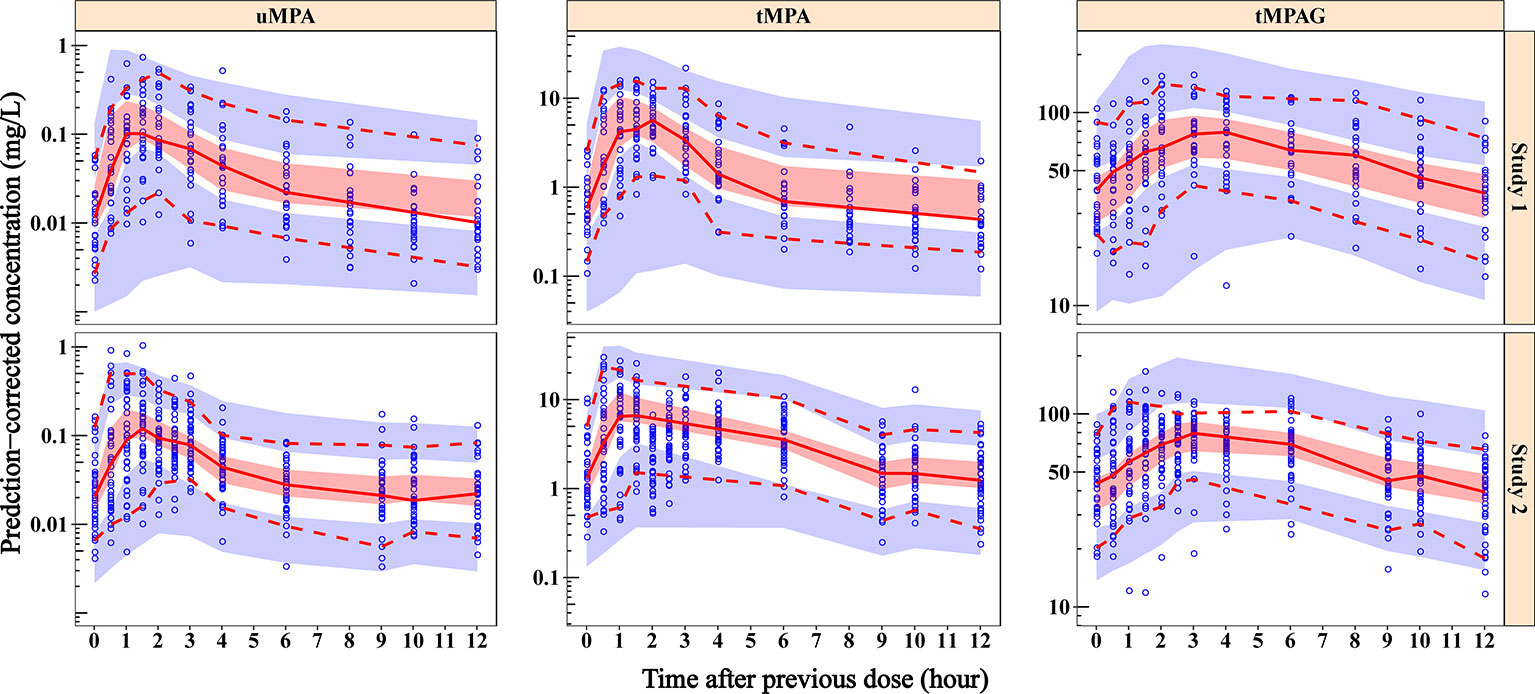
Frontiers | Effect of Protein Binding on Exposure of Unbound and Total Mycophenolic Acid: A Population Pharmacokinetic Analysis in Chinese Adult Kidney Transplant Recipients | Pharmacology

Population Pharmacokinetics of Imipenem in Critically Ill Patients: A Parametric and Nonparametric Model Converge on CKD-EPI Estimated Glomerular Filtration Rate as an Impactful Covariate | SpringerLink
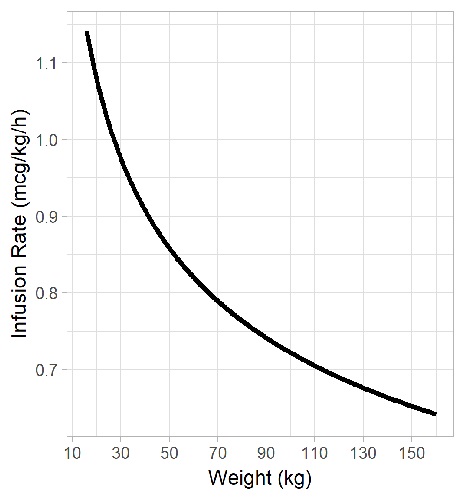
Dosing of Continuous Fentanyl Infusions in Obese Children: A Population Pharmacokinetic Analysis | PKMS Lab | UBC

Combining “Bottom‐up” and “Top‐down” Approaches to Assess the Impact of Food and Gastric pH on Pictilisib (GDC‐0941) Pharmacokinetics - Lu - 2017 - CPT: Pharmacometrics & Systems Pharmacology - Wiley Online Library
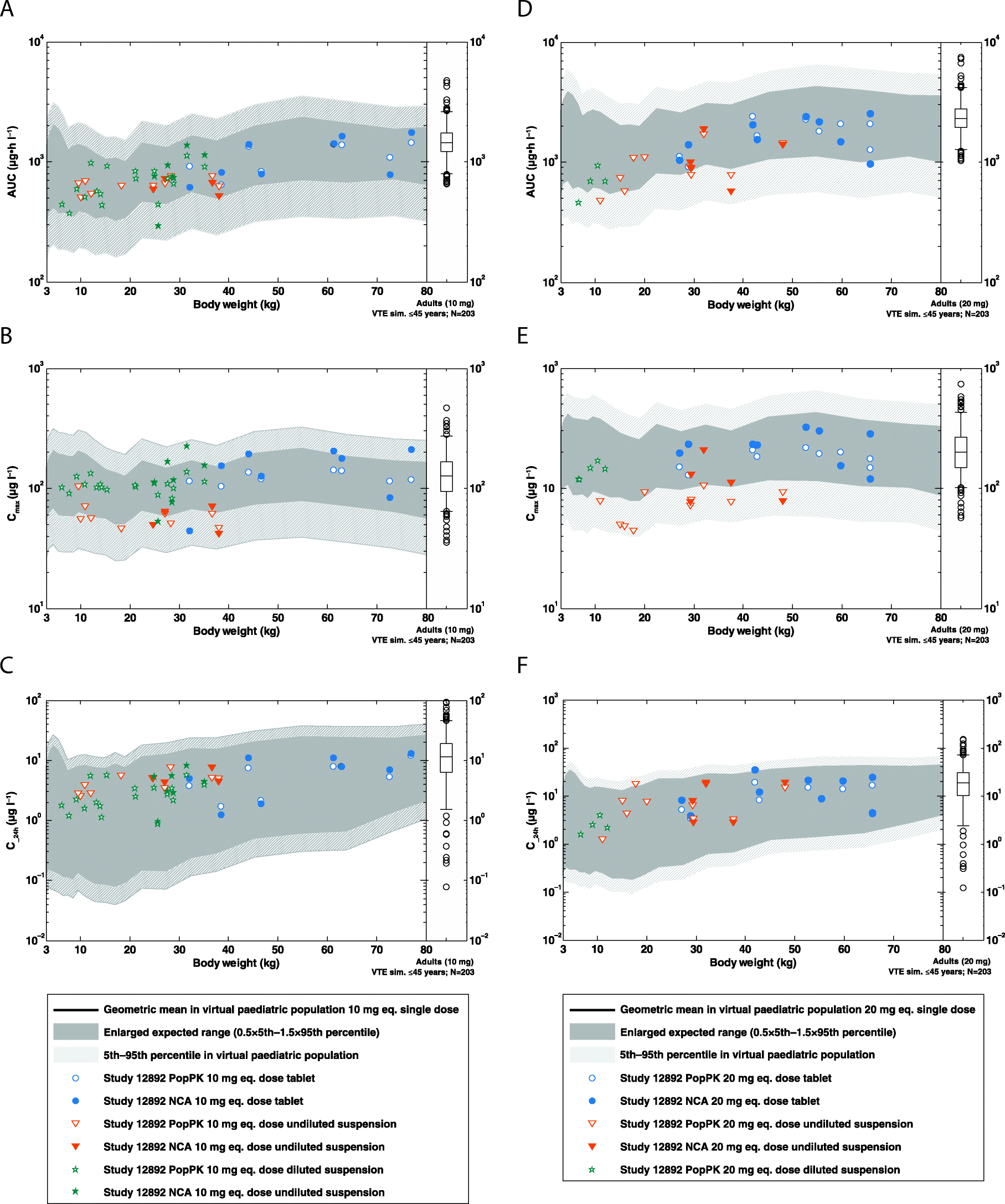
Pharmacokinetics of rivaroxaban in children using physiologically based and population pharmacokinetic modelling: an EINSTEIN-Jr phase I study | Thrombosis Journal | Full Text