European Union Medicines Agency Pharmaceutical Drug Orphan Government - Food And Administration Transparent PNG
![Community herbal monograph on Melissa officinalis .14 May 2013 . EMA/HMPC/196745/2012 . Committee - [PDF Document] Community herbal monograph on Melissa officinalis .14 May 2013 . EMA/HMPC/196745/2012 . Committee - [PDF Document]](https://demo.fdocuments.in/img/378x509/reader019/reader/2020041209/5b5db1597f8b9ac6028eea06/r-2.jpg)
Community herbal monograph on Melissa officinalis .14 May 2013 . EMA/HMPC/196745/2012 . Committee - [PDF Document]
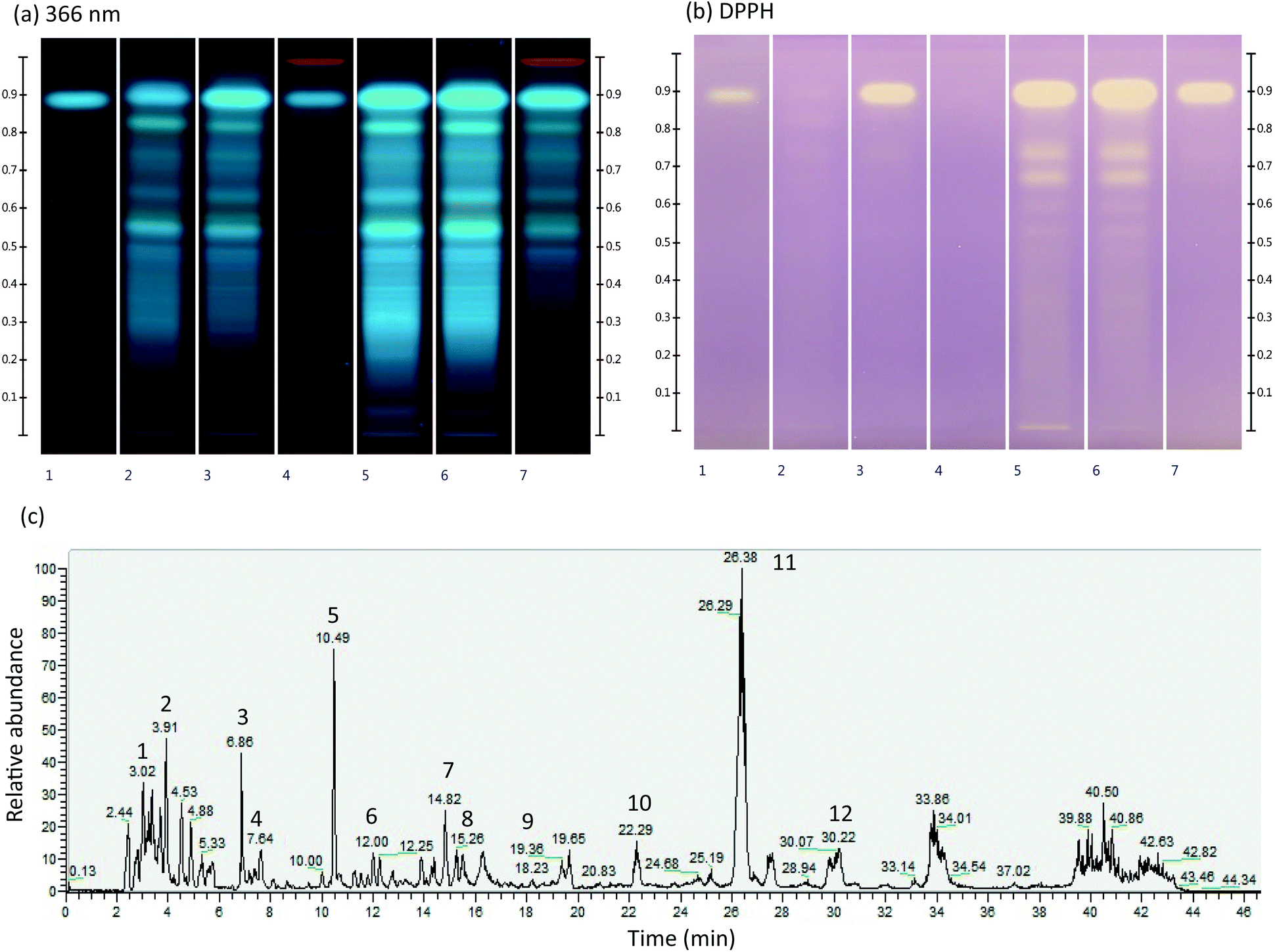
Benefits of the ipowder® extraction process applied to Melissa officinalis L.: improvement of antioxidant activity and in vitro gastro-intestinal rele ... - Food & Function (RSC Publishing) DOI:10.1039/C9FO01144G
The usage of the most frequently preferred herbal products in Turkey in nursing mothers, newborns, infants and children - Document - Gale Academic OneFile

Merck and Ridgeback Announce Initiation of a Rolling Review by the European Medicines Agency for Molnupiravir, an Investigational Oral Antiviral Medicine, for the Treatment of COVID-19 in Adults | Business Wire